 
The change in Entropy Formula is expressed as. Define the first law of thermodynamics in the context of U. 
Understand the concept of the energy units: calorie, kilocalorie and kilojoule. We will see in problem 4.11 that for a gas at room temperature and atmospheric pressure, it is appropriate to use. We can see how those high-quality sources of energy that we still have are extremely valuable, and it's critical to protect them. You need to consider the difference between the initial and final state to determine the change in entropy. First Law of Thermodynamics Second Law of Thermodynamics Learning Outcomes for Unit 4 Students will be able to. For the classical monatomic ideal gas, plot entropy as a function of particle number using both the finite size form 2.5.13 and the Sackur-Tetrode form 2.5.21. Equations relate the entropy change of a system to other properties. We can see this as the initial electrical energy from the batteries degrading into heat, which we won't be able to use or reverse into electrical energy.įrom the above, you can see that the statement: "the entropy of the universe is constantly increasing" indicates that the universe's energy is gradually degrading from high to low quality.Īt some point, the total amount of energy will be the same, but unable to generate work. From the definition of the entropy, it is known that QTdS during a reversible. However, when calculating the entropy generation, this will be positive. If we apply the first law to this system, we'll see that the energy is conserved. 0 L of water is at 99 C what is its entropy In chemical reactions involving the changes in thermodynamic quantities a variation on this equation is often. When you turn on the lamp, you'll notice the bulb is heating up, and this heat is lost to the surroundings. 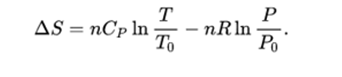
The clumped energy in the batteries is ready to be used at any time to turn on the light bulb. Enthalpy (H) and entropy (S) are related to each other by a chemical formula: G H TS where G is the change in free energy. On the other hand, we consider heat at low temperatures to be a low-quality form of energy, as this can produce little to no work.Īs a thought exercise, we can think of a battery-operated lamp. Which are forms of high-quality energy? Those that are readily available to be used, as is the case of chemical energy stored in a battery, electrical energy, mechanical energy, and some fossil fuels. This implies that we can distinguish between "high-quality" and "low-quality" forms of energy. In order to get a more intuitive idea of what entropy represents, it can be helpful to see it as a measure of the quality of the energy. In contrast to energy, entropy can feel like an abstract concept, not so simple to grasp. Up to this point, we've gone through entropy's definition, formula, and applications, but if you're still unsure what it represents, you're not alone. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
